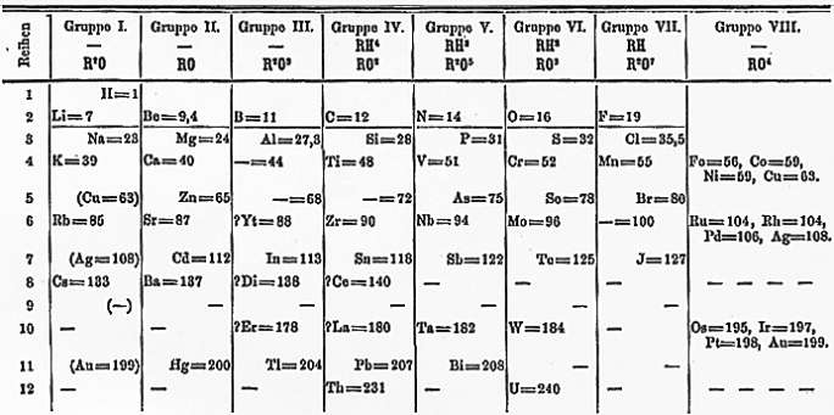
Mendeleev's impact on today's Periodic Table
He left gaps for elements he did not know and also predicted the chemical and physical properties of the unknown elements. Throughout this process, he was prepared to place elements different to the pattern if experimental evidence contradicted what he thought. Likewise, he was also willing to place the elements in the wrong order of atomic mass if common properties supported his theory of groups. Elements were generally grouped together with those with similar properties. Scientists after Mendeleev found the missing elements he had left gaps for. His prediction of their properties was correct.