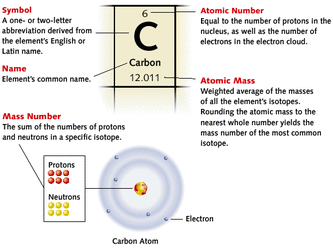
How is today's Periodic Table used / how does it affect today's society?
Thanks to the periodic table, specifically the work of Mendeleev and also the work of other scientists, we are better able to understand patterns and relationships between elements. This helps society in a variety of applications, allowing us to use the elements in mining, engineering and other science based occupations. Having the elements meticulously arranged on the periodic table also allowed us to make synthetic elements.
What does each part of the Periodic Table mean/represent?
- The columns are called groups. There are 18 groups.
- The horizontal rows are called periods. There are 7 periods.
- Group 1 (excluding Hydrogen) is known as the Alkali Metals.
- Group 2 contains the Alkali Earth Metals.
- Groups 3-12 (excluding those on separate rows) are known as the Transition Metals.
- Group 17 non-metals are called Halogens.
- Group 18 consists of the Noble Gases.
- Elements 57-71 are known as the Lanthanoids and make up the Lanthanoid Series.
- Elements 89-103 are called Actinoids and make up the Actinoid Series.
- Elements from 93 onwards are called Transuranium elements.
- Some periodic tables also specify whether an element is a metal, a non-metal, or a metalloid.
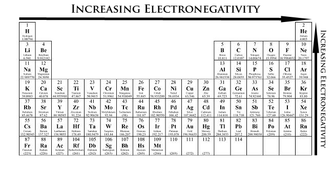
Electronegativity increases as you move to the right hand side along the periodic table (along the periods). It also decreases as you move down a group.